Terminology
/General
pH Sensor Probe vs. pH Meter: Understanding the Difference
Explore the differences between pH sensor probes and pH meters, understanding their distinct roles in measuring acidity or alkalinity.
3 min read
Advertisement
Table of Contents
In the world of chemistry, understanding the acidity or alkalinity of a solution is crucial. This is where pH comes into play. pH is essentially a mathematical tool used to measure the acidity of a solution, with values ranging from 0 to 14. You can learn more about what pH is and how it’s measured [here](link to pH explanation, if available). This article dives into the differences between two common tools used to measure pH: the pH meter and pH paper (also known as litmus paper).
A pH meter is an electronic device specifically designed to measure the pH value of a liquid. It works by gauging the concentration of H+ ions in a solution, which directly indicates its acidity or alkalinity.
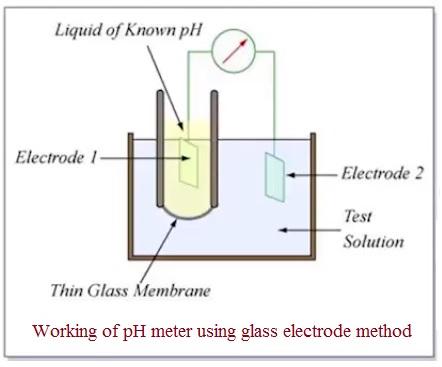
As shown in the figure above, a pH meter typically consists of two electrodes: a glass electrode (also known as the measuring electrode) and a reference electrode. These electrodes are connected to a meter capable of measuring very small voltages, usually in millivolts. The meter works by comparing the electrical potential produced by the solution being tested to the voltage of a standard solution. By measuring the difference in voltage between these two, the device can calculate the pH difference.
pH paper, or litmus paper, offers a more straightforward method for estimating pH. This paper is designed with a universal indicator, which is a mix of multiple indicators that change color depending on the pH value of the solution it comes into contact with.

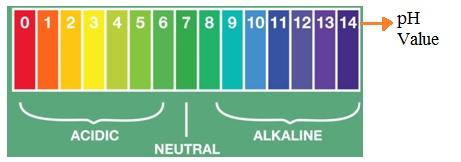
To use pH paper, you simply dip it into the liquid you want to test, leave it for a brief moment, and then remove it. The paper will then change color. This color change is then compared against a universal pH scale (shown below) to estimate the pH of the solution.
| Feature | pH Meter | pH Paper (Litmus Paper) |
|---|---|---|
| Method | Electronic measurement of voltage | Colorimetric comparison |
| Precision | Highly precise, digital readings | Approximate, qualitative readings |
| Technology | Utilizes electrodes and voltage measurement | Relies on color change of a chemical indicator |
| Ease of Use | Requires calibration and some training | Very easy to use, suitable for quick checks |
| Cost | Generally more expensive | Relatively inexpensive |
| Reading Type | Digital | Visual (color matching) |
In summary, while both pH meters and pH paper are used to measure pH, they differ significantly in their method, precision, and ease of use. A pH meter offers precise, digital readings and is essential for applications needing accurate measurements. pH paper is a simpler, less expensive option, suitable for quick, approximate checks. Depending on the application and the level of accuracy required, either tool can be the right choice.
Advertisement
Terminology
/General
Explore the differences between pH sensor probes and pH meters, understanding their distinct roles in measuring acidity or alkalinity.